Positive Clinical Results from Phase 2 Trial of SGX945 for the Treatment of Behçet's Disease Published in Rheumatology (Oxford)
- Study results support advancing SGX945 in this difficult-to-treat orphan disease
- Results suggest potential durability of response for maintenance therapy
PRINCETON, N.J., Dec. 18, 2025 /PRNewswire/ -- Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that the results from its Phase 2a proof of concept study evaluating SGX945 (dusquetide) in the treatment of Behçet's Disease have been published in Rheumatology (Oxford), in an article entitled "Results from a Pilot Study of Dusquetide for the Treatment of Aphthous Ulcers Associated with Behçet Syndrome".
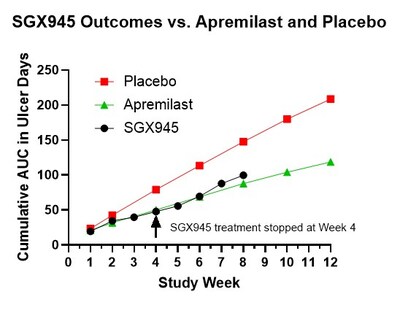
The Phase 2a study, evaluating control of oral ulcers in Behçet's Disease, reported beneficial effects for 7 of 8 patients, over the 4 weeks of treatment as well as a potentially enduring effect through the 4 weeks of follow-up. Many Behçet's Disease treatments, including the most recently approved apremilast (Otezla®), do not have an enduring impact, necessitating frequent and continuous administration. Using the Phase 3 study of apremilast as a baseline for comparison, this open-label study indicated that the area under the curve (AUC; a composite measurement of both peak number of oral ulcers and the time to resolution of the oral ulcers), average number of oral ulcers, and improvements in oral pain for SGX945 were similar to outcomes obtained in the apremilast study. Notably, outcomes in weeks 5 through 8 continued to show similar outcomes, even though apremilast treatment was continued through this period whereas SGX945 treatment was stopped at Week 4, per study design.
The primary endpoint in the Phase 3 apremilast study was the AUC of the mean number of ulcers versus time. Using this same endpoint after 4 weeks of treatment, the SGX945 treated group had a 40% improvement relative to the placebo group from the Phase 3 apremilast study, whereas apremilast had a 37% improvement relative to placebo. This improvement was sustained throughout the 4-week follow-up after treatment with SGX945, with 32% improvement evaluated at Week 8 despite treatment having stopped at Week 4. In contrast, apremilast, which was continuously administered through Week 12, had a 41% improvement at Week 8.
The improvements in oral pain mimicked the results in the AUC measurement. Seven of 8 patients reported perceived benefit with SGX945 treatment, with common outcomes including reduced duration of oral ulcers, reduced number of oral ulcers, and reduced oral pain. One patient began the study with a punctuated skin ulcer and this also resolved during the 4-week treatment with SGX945. Skin ulcers are generally considered very difficult to resolve and usually require protracted treatment. Notably, some patients also explicitly reported experiencing fewer ulcers and less pain during the 4-week follow-up period, as also reflected in the numerical analysis. SGX945 was well-tolerated with no treatment-related adverse events. Common adverse events for apremilast included diarrhea (41% of patients), nausea (19% of patients) and headache (14% of patients), none of which were observed with SGX945.
"We are pleased to publish the data from our recent SGX945 Phase 2a trial in aphthous ulcers of Behçet's Disease, enabling the medical community to evaluate this novel mechanism and therapeutic," stated Christopher J. Schaber, PhD, President and Chief Executive Officer of Soligenix. "Given the role of the innate immune system, we believe that dusquetide may offer significant relief to patients. With these, now published, results, we intend to embark on a reformulation of SGX945 to enable home-based treatment, using subcutaneous injection as used for example with weight-loss drugs. We are excited to expand dusquetide's development into different innate immune-related inflammatory conditions, such as Behçet's Disease, as a component of our long-term strategy to enhance the value of this unique compound. Behçet's Disease is an area of unmet medical need, with up to 18,000 people in the U.S., 50,000 in Europe, 350,000 people in Turkey and as many as 1 million people worldwide affected by this incurable and chronic disease. We look forward to undertaking reformulation and interacting with the health authorities in designing a follow-on clinical study."
About Dusquetide
Dusquetide, the active ingredient in SGX945 (Behçet's Disease) and SGX942 (oral mucositis), is an innate defense regulator (IDR), a new class of short, synthetic peptides. It has a novel mechanism of action whereby it modulates the body's reaction to both injury and infection towards an anti-inflammatory, anti-infective, and tissue healing response. IDRs have no direct antibiotic activity but, by modulating the host's innate immune system responses, increase survival after infections caused by a broad range of Gram-negative and Gram-positive bacterial pathogens. Dusquetide also accelerates resolution of tissue damage following exposure to a variety of agents including bacterial pathogens, trauma, and chemo- and/or radiation therapy. Preclinical efficacy and safety have been demonstrated in numerous animal disease models including mucositis, colitis, macrophage activation syndrome as well as bacterial infections. In addition, potential anti-tumor activity has been demonstrated in multiple in vitro and in vivo xenograft studies.
Dusquetide has demonstrated safety and tolerability in a Phase 1 clinical study in 84 healthy human volunteers. In Phase 2 and 3 clinical studies with dusquetide in over 350 subjects with oral mucositis due to chemoradiation therapy for head and neck cancer, positive efficacy results were demonstrated, including potential long-term ancillary benefits.
Dusquetide has also demonstrated biological efficacy and safety in a Phase 2a pilot study in 8 patients with Behçet's Disease. The Phase 2a study was an open-label study designed to be highly comparable (e.g., study endpoints, inclusion-exclusion criteria) to the published Phase 3 study which was used to support marketing approval of apremilast (Otezla®) for oral ulcers in Behçet's disease. The primary endpoint in the Phase 3 apremilast study was the area under the curve (AUC) of the mean number of ulcers versus time. Using this same endpoint after 4 weeks of treatment, the SGX945 treated group had a 40% improvement relative to the placebo group from the Phase 3 apremilast study, whereas apremilast had a 37% improvement relative to placebo. This improvement was sustained throughout the 4-week follow-up after treatment with SGX945, with 32% improvement evaluated at Week 8 despite treatment having stopped at Week 4. In contrast, apremilast, which was continuously administered through Week 12, had a 41% improvement at Week 8. One patient began the study with a punctuated skin ulcer and this also resolved during the 4-week treatment with SGX945. Skin ulcers are generally considered very difficult to resolve and usually require protracted treatment. Notably, some patients also explicitly reported experiencing fewer ulcers and less pain during the 4-week follow-up period, as also reflected in the numerical analysis. SGX945 was well-tolerated with no treatment-related adverse events. Common adverse events for apremilast included diarrhea (41% of patients), nausea (19% of patients) and headache (14% of patients), none of which were observed with SGX945.
Soligenix has a strong intellectual property position in the IDR technology platform, including composition of matter for dusquetide and related analogs. Dusquetide was developed pursuant to discoveries made by Professors B. Brett Finlay, PhD and Robert Hancock, PhD of the University of British Columbia, Canada. Dusquetide has been awarded Fast-Track designation for the treatment of oral lesions of Behçet's Disease and Orphan Drug designation for the treatment of Behçet's Disease by the FDA.
About Behçet's Disease
Behçet's Disease is commonly known as an inflammatory disorder of the blood vessels (vasculitis). Often first diagnosed in young adults, its effects and severity will wax and wane over time. Major signs and symptoms usually include mouth sores (approximately 95% of patients), skin rashes and lesions (approximately 50% of patients), genital sores (approximately 50% of patients), leg ulcers (approximately 40% of patients) and eye inflammation (approximately 15% of patients). It is a painful disease, directly impacting the patient's quality of life and ability to productively engage in life activities, including work.
Behçet's Disease is thought to be an auto-immune disease with both genetic and environmental factors. It is most common along the "Silk Road" in the Middle East and East Asia, including Turkey, Iran, Japan and China. There are approximately 18,000 known cases of Behçet's Disease in the U.S. and over 50,000 in Europe. There are as many as 1,000,000 people worldwide living with Behçet's Disease.
There is no cure for Behçet's Disease, rather treatments are prescribed to manage symptoms. Treatments may include both maintenance therapies and those specifically addressing flares (e.g., mouth ulcers, genital ulcers and leg ulcers). Corticosteroids are generally applied topically to sores and as eyedrops and may also be given systemically to reduce inflammation. Although used frequently, they have limited efficacy over the long-term and have significant side effects that become more concerning with more chronic use. Genital ulcers are often associated with significant genital scarring while leg ulcers can result in a post-thrombotic syndrome. Other treatments for Behçet's Disease flares involve suppressing the immune system with drugs (e.g., cyclosporine or cyclophosphamide). These drugs come with a higher risk of infection, liver and kidney problems, low blood counts and high blood pressure. Finally, anti-inflammatory drugs are also used, including anti-TNF medications. The only approved drug in Behçet's Disease is apremilast, which is used as a maintenance therapy to prevent formation of oral ulcers. Unfortunately, apremilast must be used continuously to be effective and is associated with both high cost and side effects including diarrhea, nausea, upper respiratory tract infection and headache.
About Soligenix, Inc.
Soligenix is a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need. Our Specialized BioTherapeutics business segment is developing and moving toward potential commercialization of HyBryte™ (SGX301 or synthetic hypericin sodium) as a novel photodynamic therapy utilizing safe visible light for the treatment of cutaneous T-cell lymphoma (CTCL). With successful completion of the second Phase 3 study, regulatory approvals will be sought to support potential commercialization worldwide. Development programs in this business segment also include expansion of synthetic hypericin (SGX302) into psoriasis, our first-in-class innate defense regulator (IDR) technology, dusquetide (SGX942) for the treatment of inflammatory diseases, including oral mucositis in head and neck cancer, and (SGX945) in Behçet's Disease.
Our Public Health Solutions business segment includes development programs for RiVax®, our ricin toxin vaccine candidate, as well as our vaccine programs targeting filoviruses (such as Marburg and Ebola) and CiVax™, our vaccine candidate for the prevention of COVID-19 (caused by SARS-CoV-2). The development of our vaccine programs incorporates the use of our proprietary heat stabilization platform technology, known as ThermoVax®. To date, this business segment has been supported with government grant and contract funding from the National Institute of Allergy and Infectious Diseases (NIAID), the Defense Threat Reduction Agency (DTRA) and the Biomedical Advanced Research and Development Authority (BARDA).
For further information regarding Soligenix, Inc., please visit the Company's website at https://www.soligenix.com and follow us on LinkedIn and Twitter at @Soligenix_Inc.
This press release may contain forward-looking statements that reflect Soligenix's current expectations about its future results, performance, prospects and opportunities, including but not limited to, potential market sizes, patient populations and clinical trial enrollment. Statements that are not historical facts, such as "anticipates," "estimates," "believes," "hopes," "intends," "plans," "expects," "goal," "may," "suggest," "will," "potential," or similar expressions, are forward-looking statements. These statements are subject to a number of risks, uncertainties and other factors that could cause actual events or results in future periods to differ materially from what is expressed in, or implied by, these statements. Soligenix cannot assure you that it will be able to successfully develop, achieve regulatory approval for or commercialize products based on its technologies, particularly in light of the significant uncertainty inherent in developing therapeutics and vaccines against bioterror threats, conducting preclinical and clinical trials of therapeutics and vaccines, obtaining regulatory approvals and manufacturing therapeutics and vaccines, that product development and commercialization efforts will not be reduced or discontinued due to difficulties or delays in clinical trials or due to lack of progress or positive results from research and development efforts, that it will be able to successfully obtain any further funding to support product development and commercialization efforts, including grants and awards, maintain its existing grants which are subject to performance requirements, enter into any biodefense procurement contracts with the U.S. Government or other countries, that it will be able to compete with larger and better financed competitors in the biotechnology industry, that changes in health care practice, third party reimbursement limitations and Federal and/or state health care reform initiatives will not negatively affect its business, or that the U.S. Congress may not pass any legislation that would provide additional funding for the Project BioShield program. In addition, there can be no assurance as to the timing or success of any of its clinical/preclinical trials. Despite the statistically significant result achieved in the first HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma or any other studies (including the open-label, investigator-initiated study) and the overall blinded study response rate observed in the second HyBryte™ (SGX301) Phase 3 clinical trial, there can be no assurance that the second HyBryte™ (SGX301) Phase 3 clinical trial will be successful or that a marketing authorization from the FDA or EMA will be granted. Additionally, although the EMA has agreed to the key design components of the second HyBryte™ (SGX301) Phase 3 clinical trial, no assurance can be given that the Company will be able to modify the development path to adequately address the FDA's concerns or that the FDA will not require a longer duration comparative study. Notwithstanding the result of HyBryte™ (SGX301) in the first Phase 3 clinical trial (or any other studies) for the treatment of cutaneous T-cell lymphoma and the Phase 2a clinical trial of SGX302 for the treatment of psoriasis, there can be no assurance as to the timing or success of the clinical trials of SGX302 for the treatment of psoriasis. Additionally, despite the biologic activity observed in aphthous ulcers induced by chemotherapy and radiation, there can be no assurance as to the timing or success of the clinical trials of SGX945 for the treatment of Behçet's Disease. Further, there can be no assurance that RiVax® will qualify for a biodefense Priority Review Voucher (PRV) or that the prior sales of PRVs will be indicative of any potential sales price for a PRV for RiVax®. Also, no assurance can be provided that the Company will receive or continue to receive non-dilutive government funding from grants and contracts that have been or may be awarded or for which the Company will apply in the future. These and other risk factors are described from time to time in filings with the Securities and Exchange Commission (the "SEC"), including, but not limited to, Soligenix's reports on Forms 10-Q and 10-K. Unless required by law, Soligenix assumes no obligation to update or revise any forward-looking statements as a result of new information or future events.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/positive-clinical-results-from-phase-2-trial-of-sgx945-for-the-treatment-of-behcets-disease-published-in-rheumatology-oxford-302645452.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/positive-clinical-results-from-phase-2-trial-of-sgx945-for-the-treatment-of-behcets-disease-published-in-rheumatology-oxford-302645452.html
SOURCE SOLIGENIX, INC.
Übrigens: US-Aktien sind bei finanzen.net ZERO sogar bis 23 Uhr handelbar (ohne Ordergebühren, zzgl. Spreads). Jetzt kostenlos Depot eröffnen und Neukunden-Bonus sichern!